Protocols
Cytospin preparation technique
Protocol created by Lorena Maestre - Monoclonal Antibodies Unit, Centro Nacional de Investigaciones Oncológicas
Cytospin preparations
A cytospin is obtained by employing centrifugal force to isolate, concentrate and deposit a monolayer of cells from a dilute cell suspension onto a circular area on a slide. The objective is to keep cells intact enabling the morphology of the cells to be examined. This technique has a wide variety of applications including cytology, histology and hematology.
Equipment, reagents and cell preparations
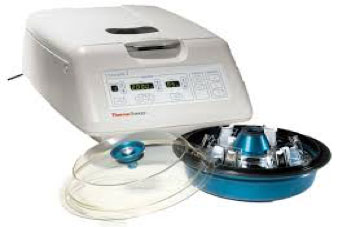
- Cytospin™ Centrifuge with cytofunnels and cytoclips (Thermo Scientific) - Figure 1
- Filter cards (Fisher Scientific 11630601)
- SuperFrost Plus Glass slides (VWR 631-0108)
- Cytofunnels (Fisher Scientific 11610591 or 11660591)
- Fixative: acetone (undiluted), methanol (ice-cold 100% methanol) or paraformaldehyde (2%) at room temperature.
- Hydrophobic pen (Vector Laboratories H-4000)
- 1% Virkon (Fisher Scientific) or bleach
- Glass pipettes
- Pencil
- Protein containing medium: RPMI 1640 medium (Sigma, R8758-500ML)+10%FBS
- Cells under study in a single cell suspension of 0.5 x 106 cells/ml in protein containing medium (e.g. RPMI1640 containing 10% foetal calf serum, Sigma)
Procedure
- Label the slides indicating the cell line used with a pencil.
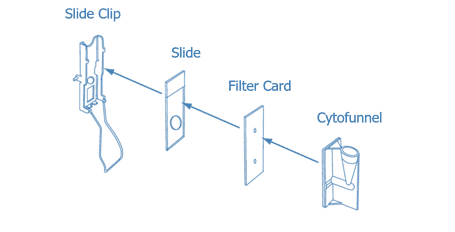
- Insert the slide into the slideclip (holder) ensuring that the frosted upper part of the slide is face up. Put the filter card (with the absorbent surface touching the slide) and position the cytofunnel next to the filter paper making sure that all the holes match up. Fasten the holder (see Image 2).
- Place the mounted holder in the corresponding recess in the Cytospin. A maximum of twelve slides can be centrifuged at once. Remember that the Cytospin must always be balanced.
- Prepare a cell suspension of not more than 0.5 x 106 cells/ml in the protein-containing medium. Note that the optimal concentration of cells may have to be determined for each cell line.
- Gently pipette the cell suspension into the cytofunnel. We recommend putting two and three drops (corresponding to 100–200µl of cell suspension, respectively) in different cytofunnels to establish the optimal volume necessary to result in a good monolayer of cells with no overlapping of cells or cell clumps. The loading volume should be optimized for each cell line used.
- Close the Cytospin lid and centrifuge at 1000 × g for 5 min.
- Remove the slides, filter cards, and cytofunnels from the centrifuge. Be careful not to move the filter paper on the slide and so damage/smudge the cell preparation.
- If desired, then the glass slides and filter cards can be removed from the cytofunnel, turned upside down before being placed with the cytofunnel and returned to the Cytospin. Cells can then be added and centrifuged to produce a slide with two cell preparations.
- Place each slide on a slide tray with the cell preparation uppermost for at least 2h at room temperature. We recommend leaving the slides overnight at room temperature to dry.
- Put the used funnels in a bucket of 1% Virkon for 30 minutes (alternative: bleach), rinse, wash and rinse in distilled water. Dry them before any further use.
- Fix the air-dried slides for 10 min in acetone at room temperature (alternatives fixatives include 10 min in 2% paraformaldehyde or 10 min in 100% methanol). The fixation method should be optimized for each experiment.
- Let the slides dry for 2h, wrap in foil or put into sealed boxes and store at -20°C until use.
- Allow the slides come up to room temperature before unwrapping or removing them from sealed boxes. This prevents problems with condensation on the slides.
- Mark the position of the cells on the slide with a hydrophobic marker. The slides are now ready for use.