Protocols
Odyssey Western Blotting protocol (OdWB)
Protocol created by Lorena Maestre - Monoclonal Antibodies Unit, Centro Nacional de Investigaciones Oncológicas
Material
- Bio-Rad Mini-Protean II Dual Slab Cell
- PowerPac power supply (100V-200mA)
- Bio-Rad Mini Trans-Blot Transfer Cell
- PVDF membrane (Millipore IPFL00010)
- Mini-PROTEAN TGX Precast Gel (Bio-Rad #4561033)
- 30% Acrilamide-Bisacrilamide solution (Bio-Rad 161-0158)
- Tetramethylethyllenediamine (1,2-TEMED - MERCK)
- 1M TrisHCl pH 8.8 (Sigma)
- 1M TrisHCl pH 6.8 (Sigma)
- Ammonium persulphate (PSA, Sigma)
- SDS 10% (Sigma)
- IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 680 (Thermo Fisher polyclonal anti mouse/rat/rabbit) Ref: A21057, A21096 and A21076)
- Tween 20 (Panreac)
- Phosphate buffered saline (PBS, homemade)
- Intercept PBS Blocking Buffer (LI-COR Biosciences, P/N: 927-70001)
- LI-COR Odyssey Imaging System (Model: CLx)
- 10X Electrophoresis running buffer (SIGMA T7777) (Electrophoresis running buffer: 10x Tris-Glycine-SDS Buffer in distilled water)
- 10X Tris-Glycine buffer (Thermo Scientific 28363) (Transfer buffer: 10x Tris-Glycine Buffer + 20% Methanol in distilled water)
- Methanol (Panreac)
- Molecular weight standard markers (Panreac)
- Loading buffer (Laemmli 2x, Sigma)
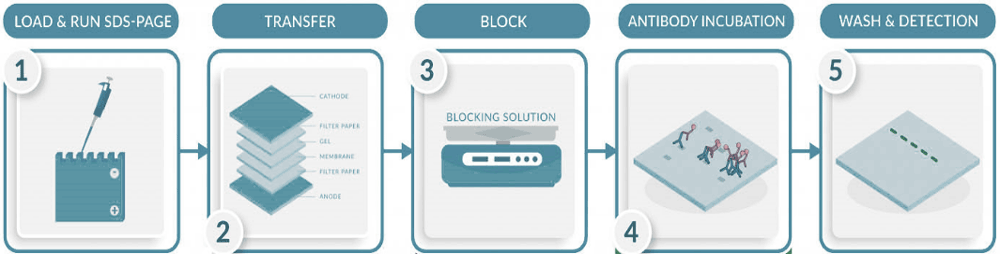
Method
- Choose the appropriate percentage precast gel and remove the top comb and bottom green plastic film or manufacture the gel according to the expected molecular weight of the antigen (Appendix I). Put the gel into the tank and fill with the electrophoresis running buffer.
- Prepare cell lysates by mixing with loading buffer as follows (10-30ug/well for transfected cells, 100-200ug/well for non- transfected cells or tissue extracts).
- Heat the samples for 5 min at 95°C, centrifuge and load each sample into individual wells of the stacking gel.
- Load 5-10ul of the molecular weight standard markers to one well.
- Connect the tank to the PowerPac power supply and run the gel at 100 V until the gel front reaches the end of the gel.
- Open the gel cassette and extract the gel, place it gently into the transfer buffer for 5 min.
- Cut the PDVF membrane to size and activate it in the blotting membrane in methanol (1min), wash in distilled water (5min) and finally equilibrate in transfer buffer (5 min).
- Prepare the blotting “sandwich” using the gel holding cassette. Remove the air bubbles by gently rolling a Pasteur pipette over the “sandwich”. Place it into the electrophoresis blotting module.
- Fill the tank with the transfer buffer and connect it to the PowerPac power supply for 1 hour at 95V (alternative: overnight at 35V, Remove the cassette and open.
- Incubate the membrane with Intercept Blocking Buffer and shake overnight at 4°C or for two hours at 37°C to block non-specific binding to the membrane.
- Incubate the membrane with the primary antibody for 1 hour at room temperature. Ensure the volume of the antibody solution is enough to fully cover the membrane. Use another primary antibody as loading control, choose one that has a different molecular weight from your protein of interest (e.g. tubulin, GAPDH, lamin, vinculin, etc). It can be done by cutting the membrane or re-staining it after the scanning.
- Wash the blotting membrane for 10 mins by shaking in PBS + 0.1% Tween-20. Repeat twice more.
- Incubate with the corresponding Alexa Fluor secondary antibody diluted 1:500 in Intercept PBS Blocking Buffer for 1 hour at room temperature.
- Wash the blotting membrane with PBS + 0.1% Tween 20 for 10 min. Repeat twice more.
- Scan the blotting membrane using the LI-COR Odyssey CLx scanner exciting the secondary fluorochrome with the correct laser.
- Adjust the bright and contrast controls to reduce the background. Save and export the file as image.
Appendix I
| Acrilamide percentage | 7.5 | 9 | 10 | 12.5 | 15 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0.75 mm gel | 1 | 2 | 1 | 2 | 1 | 2 | 1 | 2 | 1 | 2 |
| 1M TrisHCl, pH 8.8 | 1 ml | 2 ml | 1 ml | 2 ml | 1 ml | 2 ml | 1 ml | 2 ml | 1 ml | 2 ml |
| Acrilamide/Bisacrilamide | 1 ml | 2 ml | 1.2 ml | 2.4 ml | 1.33 ml | 2.66 ml | 1.67 ml | 3.34 ml | 2 ml | 4 ml |
| H2O | 1.94 ml | 3.88 ml | 1.74 ml | 3.48 ml | 1.61 ml | 3.22 ml | 1.27 ml | 2.54 ml | 0.94 ml | 1.88 ml |
| 10% SDS | 40 μl | 80 μl | 40 μl | 80 μl | 40 μl | 80 μl | 40 μl | 80 μl | 40 μl | 80 μl |
| TEMED | 5 μl | 10 μl | 5 μl | 10 μl | 5 μl | 10 μl | 5 μl | 5 μl | 5 μl | 10 μl |
| 10% PSA | 15 μl | 30 μl | 15 μl | 30 μl | 15 μl | 30 μl | 15 μl | 30 μl | 15 μl | 30 μl |
| Acrilamide percentage | 3 | |
|---|---|---|
| 0.75 mm gel | 1 | 2 |
| 1M TrisHCl, pH 6.8 | 0.5 ml | 1 ml |
| Acrilamide/Bisacrilamide | 0.2 ml | 0.4 ml |
| H2O | 1.272 ml | 2.544 ml |
| 10% SDS | 18 μl | 36 μl |
| TEMED | 2.5 μl | 5 μl |
| 10% PSA | 7.5 μl | 15 μl |
